Reversible Reactions
Normally, chemical processes involve the transformation of reactants into products, for example if we dissolve sodium metal in water, we produce an aqueous solution of sodium hydroxide and hydrogen gas. Although we don't usually say it, these are "one-way" reactions, they go from "A to B" and that's it however some reactions can go the other way and these are said to be "reversible".
Generically:

There are a number of standard GCSE level example of reversible reactions, perhaps two of the most well-known are the reaction between Ammonia and Hydrogen Chloride which produces the dense white fumes of Ammonium Chloride, and the reaction between Hydrogen and Nitrogen to produce Ammonia.
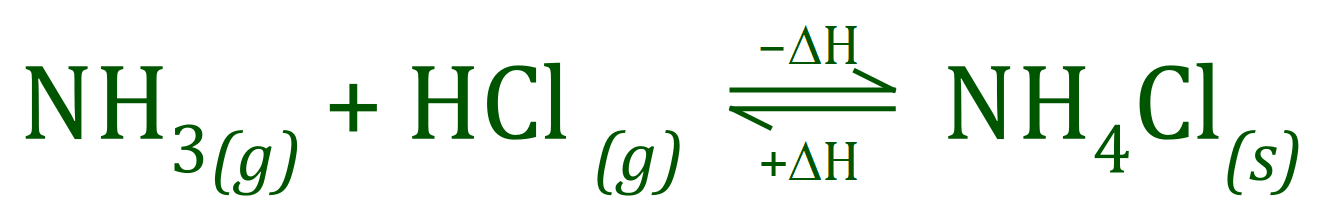
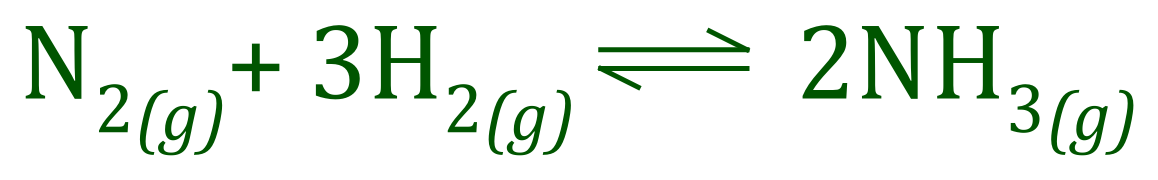
If a reversible reaction takes place in a closed system where none of the reactants or products can escape, there will come a point where the rates at which the reaction moves forward and at which the reaction moves backwards will be in "balance". At this point the reaction has reached "Equilibrium".
NB: A common misconception is that when the reaction reaches equilibrium point, there are equal concentrations of reactant and product on either side of the equilibrium, this is NOT necessarily the case.
The dehydration and rehydration of Copper Sulphate is also reversible, and in fact the rehydration is a common test for the presence of water. Copper Sulphate in its hydrated form is a beautifully deep blue colour, but the water of crystallisation can be driven off under intense heat to leave a white powdery "anhydrous" Copper Sulphate.
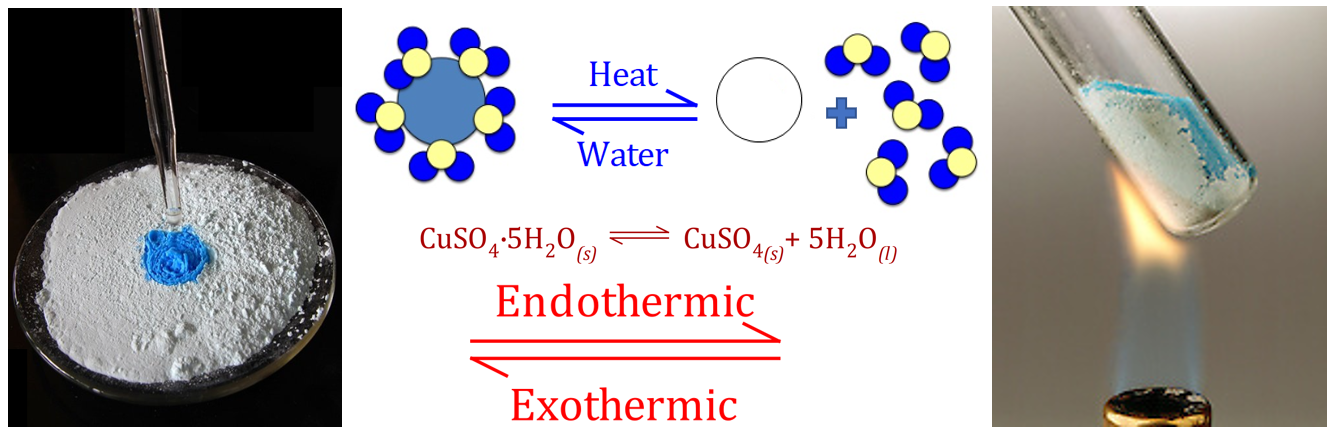
How does the reaction know which way to go? Does it go right to left? Does it go left to right? Or is there a favoured direction governed by some laws of chemistry?