Isotopes
Calculation of Relative Atomic Mass given the percentage abundances of the stable isotopes is quite straightforward if you are confident with "crunching numbers".
The sort of question you might face would be something like you were given the percentage abundance of each isotope and be asked to calculate from that the relative atomic mass. These questions are usually used to explain why elements such as chlorine appear to have "half a neutron" with a relative atomic mass of 35.5. Of course we know that this cannot be the case!
Q1 . The element Chlorine has two main, stable isotopes. Chlorine-35 with a percentage abundance of 75.77% and Chlorine-37 with a percentage abundance of 24.23%. (Chlorine does in fact have 25 isotopes ranging from Chlorine-28 to Chlorine-52 but these do not concern us because they are so short lived and of interest to theoreticians only). Using the information given for the two stable isotopes, calculate the relative atomic mass of chlorine to its usual 1 decimal place.
Chlorine seems to be a favourite with examiners for this type of calculation, let's see how to do it.
There are 2 ways to attack this question, keep the percentages as they are and then divide the final answer by 100, or convert the percentages to decimals before you perform any calculations. I've mentioned previously in this topic that I prefer the second method, and although it may not be quite as straightforward as the first method it will come in useful for the second type of calculation that I will show you subsequently. So this is the method that I will adopt.
Chlorine-35 has a percentage abundance of 75.77%, which when converted as a decimal becomes 0.7577 (we simply divide by a hundred). By the same reasoning then, Chlorine-37 will have an abundance of 0.2423 as a decimal. Stop at this point if you're not sure where these figures came from, don't carry on until you're happy.
Now that we have our percentage abundance is represented as decimals, we simply multiply each "decimal abundance" by the atomic mass that it represents and then add up the results. This will give us a relative atomic mass of the element:
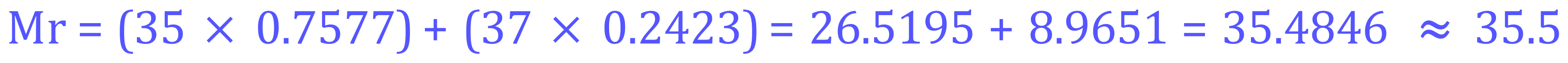
Q2. The element Uranium has three main isotopes, Uranium-234 (0.0054%), Uranium-235 (0.72%) and Uranium-238 (99.274%). Using the percentage abundance data given, calculate the relevant atomic mass of Uranium.
This question is not really any different from the previous one, we are just looking at a different element with different percentage abundances. The arithmetic is identical. We have a percentage abundances of the 3 isotopes of uranium and we convert them to decimals by dividing each by 100.
Uranium-234 (0.0054%) - 0.000054
Uranium-235 (0.72%) - 0.0072
Uranium-238 (99.274%) - 0.99274
Let's now put them into the same calculation and come up with an answer for the relative atomic mass of uranium.


From the result of this calculation we can see that the predominant isotope is in fact Uranium-238 however my periodic table shows it as 238.029. The answer that you arrive at will of course depend on where the percentage abundance values were obtained from, and many sources will differ slightly.
Q3. Hydrogen exists in three main isotopes, Hydrogen-1 (protium), Hydrogen-2 (deuterium) and Hydrogen-3 (tritium). The percentage abundances of protium equals 99.98%, deuterium equals 0.0156% and tritium equals 0.0044%. Given this data calculate the relative atomic mass of Hydrogen.
We treat this question no differently to any of the others, we are given the percentage abundances of the 3 main isotopes of hydrogen and be convert them to decimals, as we did before, by dividing each value by 100.
Hydrogen-1 (99.98%) - 0.9998
Hydrogen-2 (0.0156%) - 0.00156
Hydrogen-3 (0.0044%) - 0.000044
Our calculation therefore becomes:

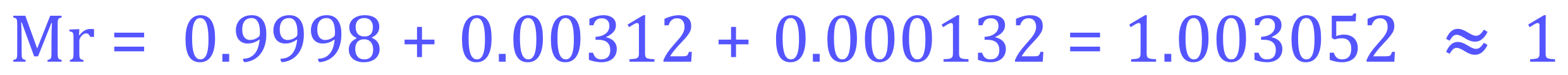
Once again, my periodic table states this as 1.008 which in real terms is only slightly out, the reason that my calculation shows 1.003 has probably got a lot to do with the possible inaccuracy of the percentage abundance data obtained. For the purposes of this exercise I only want to make sure that you can do the calculations, the data that you are provided will lead to a specific answer and will be marked accordingly.
Q4. The Group 1 Alkali Metal Potassium exists in 3 main stable isotopes, Potassium-39 at 93.3%, Potassium-40 at 0.012% and Potassium-41 at 6.688%. From this information, calculate the relative atomic mass of Potassium.
This question is identical to the previous ones so I won't explain it, I am just going to go straight to the result, see if you can do the same.

My periodic table states this as 39.098, which to one decimal place is 39.1. My calculation is also 39.1 to one decimal place.
Given the equation:

Where Mr is the relative atomic mass of the element, M1 and M2 are the atomic masses of the isotopes and x / 1-x are algebraic representations of the relative abundances for a 2 isotopes system, attempt the following questions.
Q5. The relative atomic mass of the element Nitrogen is 14.007. Nitrogen exists (for the purposes of this question) in 2 isotopic forms, Nitrogen-14 and Nitrogen-15. Using the algebraic expression above, calculate the percentage abundances of these 2 isotopes.
From the equation above, and substituting known values we can produce the following expression:
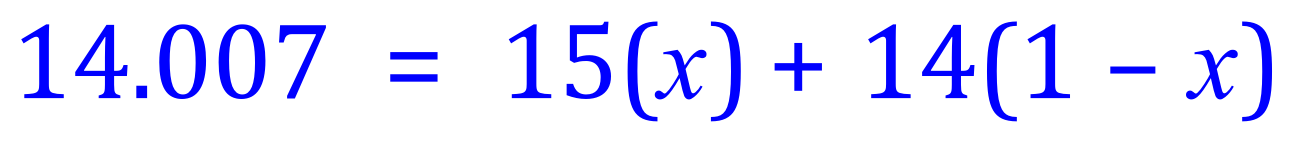
To complete the question and establish a value for 'x' we need to rearrange the above expression in terms of 'x', this is where your algebraic and mathematical dexterity will come in very useful. It would have been just as easy to say 14 (x) +15 (1 - x) because the algebraic rearrangement would produce the same result anyway, but I've chosen to do it this way so let's carry on.
Step 1 - Multiply out the brackets on the right-hand side:
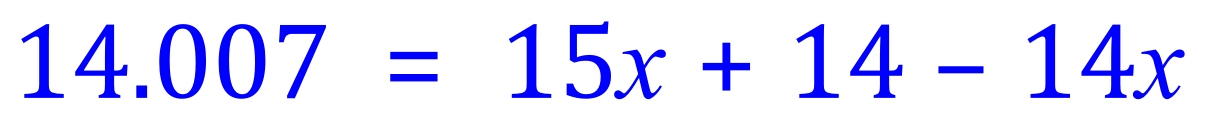
Step 2 - Tidy up the 'x' terms:
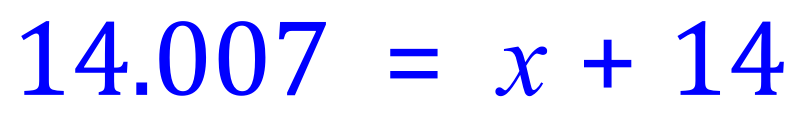
Step 3 - Rearrange this expression in terms of 'x'

Remember that this result 0.007 is the decimal equivalent of the percentage abundance of the isotope that we chose to assign to 'x', that is Nitrogen -15 so we need to multiply this value now by 100 to get it as a percentage.
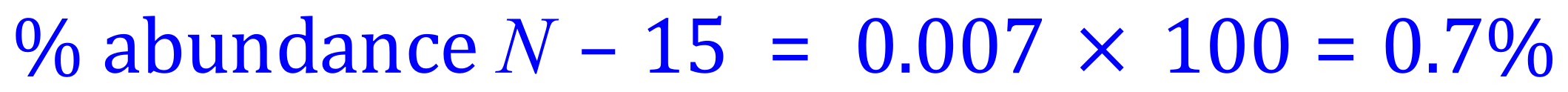
You should now be able to see that the abundance of Nitrogen-14 would be 1 - x, or 0.993. If we then multiply this by 100 we come out with a percentage abundance of 99.3% for Nitrogen-14.
Q6. The 2 main isotopes of the element Copper are Copper-65 and Copper-63, with atomic masses of 64.9278 (Copper-65) and 62.9296 (Copper-63). Given that the relative atomic mass of copper is 63.546, use the information given to calculate the percentage abundance of each isotope.
We remain with the algebraic expression that we saw above in the completion of this calculation, and we allocate one of the isotopes to the variable 'x' and the other to 1 - 'x'.

Let us say that M1 will be the atomic mass of Copper-65, and M2 the atomic mass of Copper-63, our expression should now look something like this:
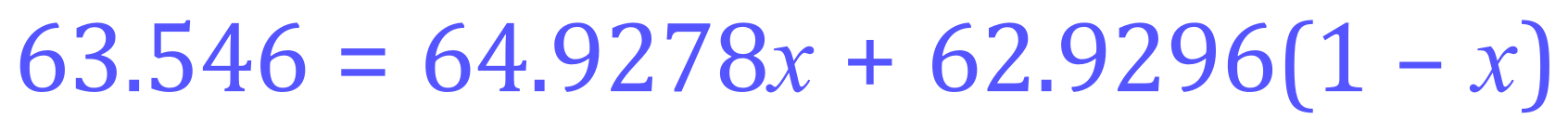
Step 1 - as before, expand the parentheses:
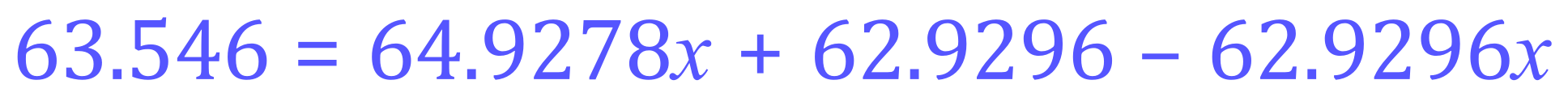
Step 2 - tidy up the 'x' terms:

Step 3 - rearrange in terms of 'x':
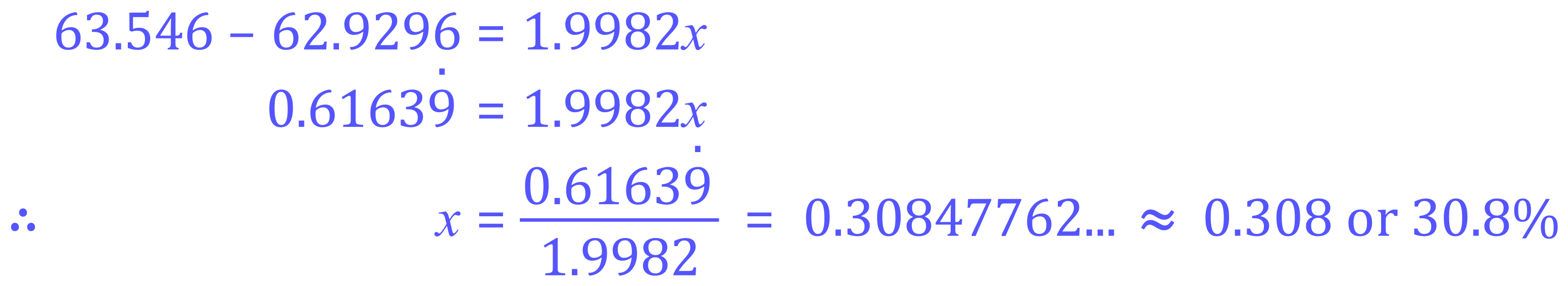
Given that we allocated the variable 'x' to the isotope Copper-65 we can now state that the percentage abundance of this particular isotope is 30.8% (Remember that the value for 'x' is a decimal representation and must therefore be multiplied by one hundred to return it to a percentage), the abundance of the other isotope (Copper-63) would therefore be 100-30.8 = 69.2%
Back to >> Questions <<
Back to >> Isotopes <<